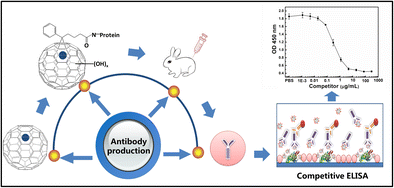
Developing an Immunoassay
The secret to creating an immunoassay for small molecular compounds and ensuring precise measurements is to generate antibodies with high affinity and selectivity, particularly when biological concentrations in patient samples are at trace levels.
Antibodies have been commonly used as the key components in diagnostic assays and utilized in several techniques for food and environmental testing as well as biohazard detection. Because of its adaptability to various types of samples and ease of service, the immunoassay is the most used rapid-detection method available today.
Antibody Application in Immunoassays
Immunoassays are fast and precise in the results they produce that can be used both remotely on-site in the field and in the lab to identify molecules of concern. Immunoassays rely on an antibody’s intrinsic ability to bind to a particular structure of a molecule known as the epitope.
Antibodies are proteins that animals manufacture naturally in response to the entry of a foreign molecule (antigen) into the body. Antibodies are present in blood and fluids surrounding tissue, and they bind to antigens whenever they meet them. The exact location the antibody binds the antigen is called the epitope.
Antibodies are highly selective and can bind only to the specific three-dimensional structure of an epitope, or analyte when they are developed to that structure. There are four typical enzyme-linked immunosorbent assay (ELISA) formats: antigen-down immunoassays, competitive inhibition assays, monoclonal-polyclonal sandwich assays, and rapid assays.
Antigen-down Immunoassays
An antigen-down ELISA uses an analyte (instead of an antibody) coated onto a 96-well microtiter plate to bind the antibodies contained in a specimen.
When a sample (such as human serum) is applied, antibodies from the sample bind to the antigen on the microtiter plate, which is then stored in the well. Following that, a species-specific antibody labeled with HRP is attached, which links to the antibody bound to the antigen on the microtiter plate.
Antigen-down assays can be formulated as rapid tests and are often used to identify allergy disorders – a patient’s blood is regularly screened against various allergens to see whether the individual has antibodies to the allergen.
Rapid Immunoassay
Rapid experiments rely on antibodies to bind to antigens and can be designed as competitive inhibition formats, antigen-down formats, or mAb-pAb sandwich formats. The antibody and antigen reagents are attached to porous membranes in a rapid examination, which reacts with positive samples when channeling excess fluids to the non-reactive portion of the membrane.
Rapid immunoassays are usually categorized into two types: lateral flow experiments, in which the sample is simply put in a well and the results are read instantly, and flow-through processes, in which the sample is placed in a well, cleaned, and then an analyte-colloidal gold conjugate is applied, and the results are read after a few minutes.
Per strip or cassette, one sample is checked. Rapid tests are used in the field by non-laboratory people checking entire samples because they are simpler than microtiter plate assays, require less sample preparation, are often easier, and produce yes/no responses without the use of an instrument.
Rapid immunoassays, on the other hand, are not as sensitive and cannot be used to reliably measure an analyte.
Monoclonal-Polyclonal Sandwich Immunoassay
A monoclonal antibody is adsorbed onto a plastic microtiter plate in a standard microtiter plate sandwich immunoassay. When the reference sample is attached to the plate, the antibody binds the target antigen from the sample and retains it in the plate.
When a polyclonal antibody is inserted in the following step, it binds to the target antigen (which is already attached to the monoclonal antibody on the plate), resulting in an antigen “sandwich” between the two antibodies.
This binding reaction can then be determined using radioisotopes or enzymes attached to the polyclonal antibody. The radioisotope or enzyme produces a radiation or color signal equal to the amount of target antigen in the initial sample applied to the tray.
The degree of color can be observed and determined for the naked eye (as with a home pregnancy test), a scintillation counter (for an RIA), or a spectrophotometric plate reader, depending on the immunoassay format (for an EIA).
Competitive Inhibition Immunoassay
Many immunoassays are arranged in a competitive inhibition format. Since competitive inhibition assays only need the binding of one antibody rather than two in typical ELISA formats, they are commonly used to quantify small analytes.
A sandwich assay format may not be possible due to the high likelihood of steric hindrance arising as two antibodies try to bind to a small molecule at the same time. As a result, a competitive inhibition assay is preferable.
For more information regarding the subject matter, contact Green Mountain Antibodies for a full range of services for creating new monoclonal antibodies or producing antibodies: 802-865-6230






